On the day Christina Tryphena thought she had recovered from COVID-19, she lost her ability to sleep.
After enduring three consecutive nights without rest, she was admitted to the emergency room. She was administered a sedative in the form of benzodiazepines, which finally allowed her to sleep for several hours. Today, she still struggles to sleep for more than three to four hours per night.
Tryphena, a 29-year-old crisis counselor and research psychologist, has post-acute sequelae of COVID-19 — more commonly known as Long COVID. The condition is a widespread complication of COVID-19 that can cause hundreds of debilitating symptoms, including intense fatigue, post-exertional malaise, and insomnia.
“I have no idea how I made it through,” Tryphena said. “My career is suffering, my academics have suffered, and I have to spend a lot of time at home.”
Although more than four years have passed since the outset of the pandemic, the lingering effects of the virus have had catastrophic consequences on the lives of millions internationally. Over 17 million people suffer from Long COVID in the United States alone. A quarter of them reported that their activities have been severely limited due to their condition.
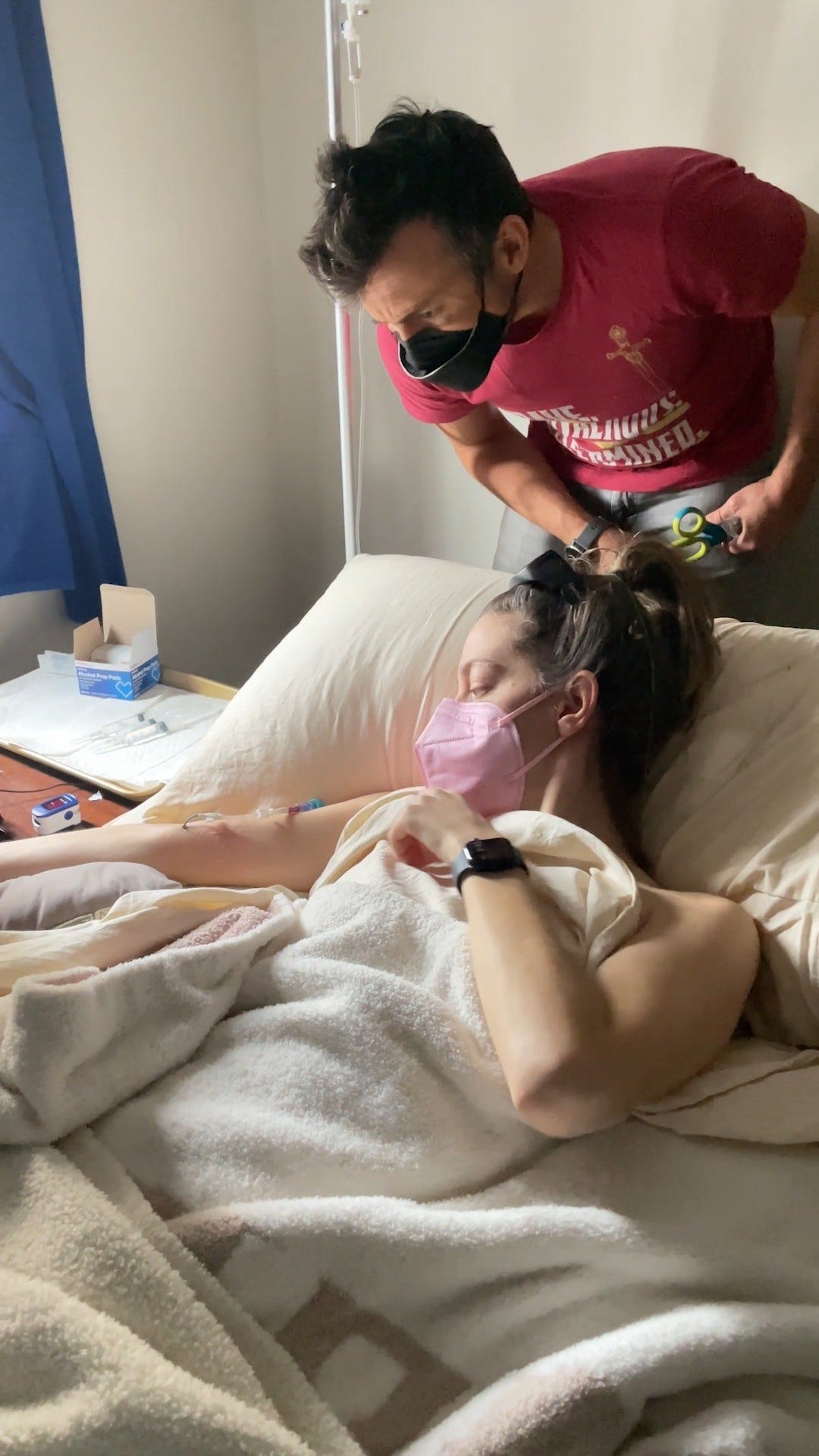
Tryphena falls into this category, as she was extremely active prior to her infection — regularly working 80 hours a week. Now, she can only complete a fraction of the work due to her insomnia. Additionally, she has abandoned hobbies such as writing and cooking because they have become too draining. Anxiety and adrenaline spikes have become frequent, which can cause her to experience severe panic attacks. Given her symptoms, she was forced to adopt a more insular lifestyle.
“As a childhood cancer survivor, I am used to being homebound and a hermit,” Tryphena said. “While this is a big stray from the trajectory I was on, I have developed quite a bit of acceptance of the situation.”
Unfortunately, there is no single treatment or test for Long COVID. Instead, patients are diagnosed by judging how closely they meet certain criteria, which can vary among medical institutions. The National Academies of Science, Engineering, and Medicine recently defined Long COVID as an “infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least three months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems.”
The issuance of a universal definition represents a milestone for the illness, which was first reported by patients on social media in 2020. In the span of several months, Long COVID transformed from a hashtag into a condition recognized by institutions such as the World Health Organization.
The estimated national financial impact of Long COVID is $3.7 trillion — on par with the Great Recession. Despite the severity of Long COVID, only half of Americans believe that investing in research and treatment is “very important”, according to a poll by the Pew Research Center.
Scientists have not yet found a definitive explanation for Long COVID, but there are several hypotheses. For example, it is possible that COVID-19 may reactivate dormant viruses in the body. This may explain Tryphena’s case, as she previously suffered from postural orthostatic tachycardia syndrome (POTS) and mast cell activation syndrome (MCAS).
These conditions can cause symptoms such as fatigue and sleep disturbances. However, they were in remission until her infection with COVID-19. Other explanations for Long COVID include organ damage and remaining coronavirus in the body provoking an immune response, leading to chronic inflammation.
Unfortunately, patients may have to wait years for a cure. Almost 600 federally funded studies on Long COVID have been completed or are ongoing, but few have yielded promising results. Among the treatments that are currently being tested are antiviral medications, antidepressants, cannabis, qigong, and probiotics. Even if a treatment is identified, it will still take considerable time before it is available to patients due to the prolonged nature of clinical trials. Until then, those affected can only manage their symptoms.
“I think things will improve, but maybe not for another decade when better understanding is gained with this condition through rigorous research,” Tryphena said. “We need more doctors and researchers allocated to studying this alone, and that may not happen as COVID has been accepted as a fact of life.”
Solving Long COVID
Part of the reason that progress in Long COVID research has been slow is that caring for hospitalized patients took priority during the early days of the pandemic.
"Patient-made evidence in the form of one case study was important when no peer-reviewed articles had yet documented long-lasting symptoms,” said Dr. Felicity Callard and Dr. Elisa Perego, in a paper. “Non-hospitalized patients were — at that point — literally beyond the gaze of biomedical research.”
As serious cases of COVID-19 decreased over time, scientists were able to dedicate more resources to Long COVID. However, researchers still have to grapple with the immense variability in how the condition presents itself in different people. With over 200 possible symptoms, it can be difficult to choose a particular drug or region of the body to investigate.
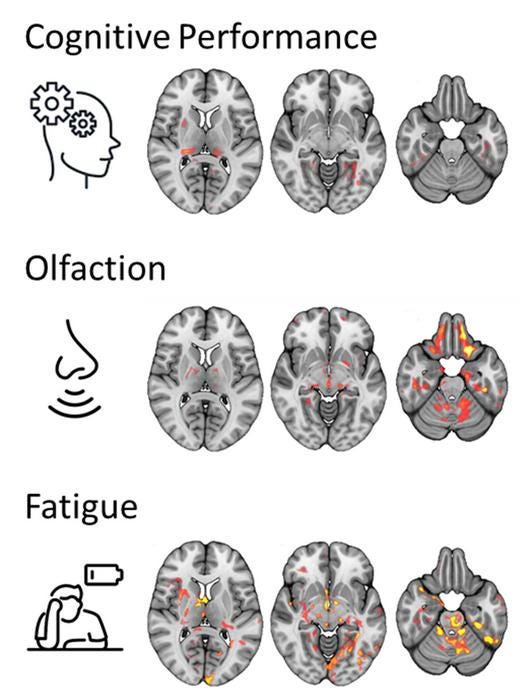
Some scientists have found promising results by utilizing new technology. For instance, a study published in November 2023 revealed that the brains of Long COVID patients had small but significant changes compared to healthy controls. The researchers used a novel technique called diffusion microstructure imaging, which allows them to discern much smaller details than a traditional MRI.
On the flip side, old drugs are being repurposed in an attempt to treat Long COVID. Several months ago, a group of researchers in Florida found promising results by administering low-dose naltrexone to patients. Naltrexone, invented in 1963, is normally prescribed to treat opioid and alcohol dependence.
However, in low doses, it can help decrease inflammation in the brain caused by Long COVID. Because the study is not a clinical trial, the research is far from definitive. Dr. Jarred Younger, the director of the Neuroinflammation, Pain, and Fatigue Lab in Alabama, acknowledged the limits of the study in a video:
This is not a clinical trial … we don’t know that it’s the treatments for sure that made them feel better. … If I had Long COVID, I would probably print this paper, I would go to my primary care physician, and I would say I am struggling with this long COVID. No one seems to know the best treatment; I would really like to try this low-dose naltrexone and give it a shot for three months. It seems to have very low side effects and it’s inexpensive.
Without a cure, those with Long COVID often perform research themselves and resort to self-treatment. For example, Tryphena was able to manage her symptoms by limiting her intake of foods high in histamine, which can trigger an immune response and cause insomnia. Many patients continue to use social media to share their experiences and chat with others facing similar issues. On Reddit, a community called “covidlonghaulers” has almost 60,000 members.
The top post is simply titled “Suicide Prevention and Support Thread”.
A familiar case
One of the positives of Long COVID is that the condition can help bring attention to similar ailments such as myalgic encephalomyelitis (ME/CFS) — also known as chronic fatigue syndrome. Like Long COVID, ME/CFS can cause fatigue, sleep and memory problems, and other issues. The two have significant overlap in terms of symptoms and severity, but research about ME/CFS has historically been underfunded.
With roughly 3 million people suffering from ME/CFS in the United States, the federal research budget is only $5 per patient. By comparison, HIV/AIDS receives almost $2,600 per patient. According to a paper, funding for ME/CFS “would need to increase roughly 14-fold” in order to match its disease burden, which is a method of quantifying the impact of a health problem.
Although it is difficult to find official explanations for why this is the case, the answer may lie in its history. In the late 1800s, ME/CFS was thought to be the result of the stressful demands of the industrial era. Almost 170 years later, the WHO classified it as a neurological issue. Still, some doctors maintained an outdated view of the condition.
When an outbreak in 1970 caused over a hundred people to develop ME/CFS, two psychiatrists faced backlash for terming it “mass hysteria” without speaking to any of the patients. Much of the criticism came from medical professionals who had fallen ill themselves.
Interestingly, this parallels how Long COVID patients originally cited first-hand accounts of doctors who had developed Long COVID to prove the graveness of their situation. In the 1980s, ME/CFS finally gained national attention when it was covered by the Hippocrates magazine, referred to as “Raggedy Ann Syndrome”.
It is possible that scientists today are discouraged by the historical connotation of ME/CFS being entirely psychological as opposed to a physiological issue. In addition, it has proved to be a difficult condition to research. In a Time article, Dr. Steven Philips and Dr. Michelle Williams argued that ME/CFS research has been “singularly unproductive”, and that it has a “low ultimate likelihood of helping patients”. They drew a parallel to the state of Long COVID research:
Research still has a vital role in the new ME/CFS/Long COVID paradigm. But it should be a different kind of research. The kind that no longer focuses on biomarkers and mechanisms. These are sure to provide “promising” but false leads and divert resources. Focus should be on health services research and on measures that directly impact the welfare of Long COVID sufferers: prevention, improved prognosis, access to empathetic care and quality of life issues.
Philips and Williams effectively want scientists to focus on symptom management as opposed to investigating the causes of these conditions, which they believe may lead to a dead end. Like with Long COVID, many patients suffering from ME/CFS are not satisfied with this approach, thus relying on their own research to find a cure. Jennifer Brea, a filmmaker, suffered for ME/CFS for eight years before receiving treatment that caused her symptoms to go into remission.
“We experimented on ourselves,” Brea said, in a TED talk.
She was eventually diagnosed with craniocervical instability and tethered cord syndrome. Once these issues were corrected with surgery, her ME/CFS began to fade. Her case may represent hope for those suffering from ME/CFS and Long COVID, as it proves that fixing biological problems can resolve seemingly irreparable conditions.


However, not all patients have access to the resources she did. Without having given her talk, Brea likely would not have met the surgeons who helped her identify the roots of her condition. Due to the sheer complexity of these conditions and the lengthy nature of research, the future remains uncertain for the vast majority of victims of chronic conditions such as Long COVID and ME/CFS.
If history is any indicator, patients will play a crucial role in driving the future of research through painstaking self-advocacy. In Brea’s words:
We need to listen to patients’ stories. We need to be willing to say “I don’t know”. “‘I don’t know” is a beautiful thing. “I don’t know” is where discovery starts.
View our sources for this piece here.





